Diethanolamine |
|||||||||||
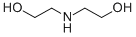 |
|||||||||||
Item : 111-42-2 |
|||||||||||
Details :Diethanolamine
Chemical Formula: HN(CH2CH2OH)2 Synonyms:2,2'-Iminodiethanol (DEA)
Packing : 220 kg, iron drum Boiling Point :269.1℃ Melting Point : 28.0℃ Identification of 111-42-2 Name: Diethanolamine (Related Reference) EINECS: 203-868-0 Molecular Formula: C4H11NO2 CAS Registry Number: 111-42-2 Synonyms: 2,2-Iminodiethanol; diolamine; Bis(beta-hydroxyethyl)amine; DEA; 2,2'-Iminodiethanol InChI: InChI=1/C4H11NO2/c6-3-1-5-2-4-7/h5-7H,1-4H2 Molecular Structure: This structure is also available as a 2d Mol fileChemical Properties Appearance: solid or viscous liquid with an amine odour Molecular Weight: 105.14 Density: 1.096 Boiling Point: 268℃ Melting Point: 27-30℃ Flash Point: 179℃ Storage Temperature: Store at RT Refractive index: 1.475-1.479 Solubility: Miscible Stability: Stable. Incompatible with carbon dioxide, strong acids, strong oxidizing agents. Deliquescent.Safety Data of 111-42-2 Risk Codes: R22;R38;R41;R48/22 Safety Statements: S26;S36/37/39;S46 Diethanolamine, often abbreviated as DEA or DEOA, is an organic compound with the formula HN(CH2CH2OH)2. Pure diethanolamine is a white solid at room temperature, but its tendency to absorb water and to supercool[1] mean it is often encountered as a colorless, viscous liquid. Diethanolamine is polyfunctional, being a secondary amine and a diol. Like other organic amines, diethanolamine acts as a weak base. Reflecting the hydrophilic character of the alcohol groups, DEA is soluble in water. Amides prepared from DEA are often also hydrophilic.
Production[edit]The reaction of ethylene oxide with aqueous ammonia first produces ethanolamine:
which reacts with a second and third equivalent of ethylene oxide to give DEA and triethanolamine:
About 300M kg are produced annually in this way.[2] The ratio of the products can be controlled by changing thestoichiometry of the reactants.[3] Uses[edit]DEA is used as a surfactant and a corrosion inhibitor. It is used to remove hydrogen sulfide and carbon dioxidefrom natural gas. In oil refineries, a DEA in water solution is commonly used to remove hydrogen sulfide from sour gas. It has an advantage over a similar amine ethanolamine in that a higher concentration may be used for the same corrosion potential. This allows refiners to scrub hydrogen sulfide at a lower circulating amine rate with less overall energy usage. DEA is a chemical feedstock used in the production of morpholine.[2][3] Amides derived from DEA and fatty acids, known as diethanolamides, are amphiphilic. Commonly used ingredients that may contain DEA[edit]DEA is used in the production of diethanolamides, which are common ingredients in cosmetics and shampoos added to confer a creamy texture and foaming action. Consequently, some cosmetics that include diethanolamides as ingredients may contain traces of DEA.[citation needed] Some of the most commonly used diethanolamides include: Safety[edit]DEA is a potential skin irritant in workers sensitized by exposure to water-based metalworking fluids.[4] One study showed that DEA inhibits in baby mice the absorption of choline, which is necessary for brain development and maintenance;[5] however, a study in humans determined that dermal treatment for 1 month with a commercially available skin lotion containing DEA resulted in DEA levels that were "far below those concentrations associated with perturbed brain development in the mouse".[6] In a mouse study of chronic exposure to inhaled DEA at high concentrations (above 150 mg/m3), DEA was found to induce body and organ weight changes, clinical and histopathological changes, indicative of mild blood, liver, kidney and testicular systemic toxicity.[7] A 2009 study found that DEA has potential acute, chronic and subchronic toxicity properties for aquatic species.[8] |
|||||||||||
| [Next51745-87-0] [Previous67-56-1] | |||||||||||
|


